Heidelberg Engineering partner RetInSight secures the first certification under EU Medical Device Regulation for clinical use of GA Monitor based exclusively on SPECTRALIS images
Heidelberg, Germany – Heidelberg Engineering is pleased to announce that Heidelberg AppWay, the first vendor neutral PACS offering a secure gateway to multiple artificial intelligence apps, now offers access to the first AI-based GA analysis algorithm approved under the EU medical device regulation for clinical use.
Vienna-based RetInSight, a developer of AI-based medical software solutions, just received approval for its GA Monitor as a Class II medical device according to the EU Medical Device Regulation 2017/745 (MDR).
The GA Monitor analyzes SPECTRALIS optical coherence tomography (OCT) images to detect and monitor geographic atrophy (GA), a late stage, non-exudative form of age-related macular degeneration (AMD) and one of the most common causes of visual impairment and blindness.
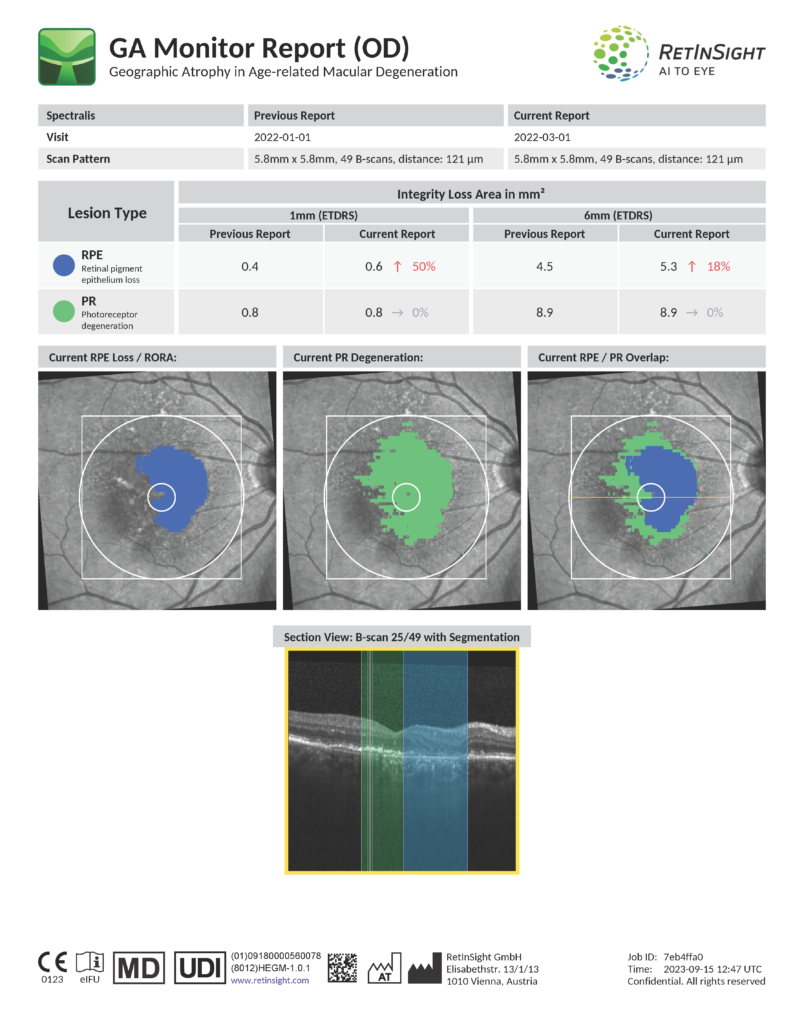
image courtesy: RetInSight GmbH
The unique feature of the RetInSight GA Monitor is its capability to visualize and measure the condition of photoreceptors. In GA, the progressive and irreversible deterioration of vision begins with photoreceptor thinning and loss, which is exceedingly difficult for ophthalmologists to detect on OCT images. Specially trained AI-supported algorithms can identify the degree and extent of the thinning of the photoreceptors (PR), as well as the loss of the retinal pigment epithelium (PE). These can help physicians identify patients in need of treatment on their first visit and track their disease progression by accurately measuring PR degeneration and RPE loss.
“With the launch of our GA Monitor, we are supporting ophthalmologists in clinical decision making by offering reliable software based on medical findings,” says Corinna zur Bonsen-Thomas, CEO of RetInSight GmbH. “The unique AI-based visualization of the photoreceptor layer sets us apart from other solutions and has the potential to fundamentally change the way ophthalmologists diagnose and treat GA patients. We are proud to be able to help preserve the vision and quality of life of millions of patients.”
GA is more prevalent with age, affects around five million people worldwide, and accounts for 35% of blindness caused by AMD.
Considering that two new therapies for the treatment of GA have already been approved in the USA and that therapeutic options are subsequently expected in Europe, patient numbers will increase significantly, requiring effective clinical management.
“These advanced therapeutics demand precision diagnostics,” says Krysten Williams, Head of Global Marketing and Education with Heidelberg Engineering. “We are dedicated to supporting confident decision making and efficient workflow in clinical routine – first with the exceptional quality of multimodal images from SPECTRALIS and now the secure access to deeper diagnostic insight provided by the RetInSight GA Monitor through HEYEX 2 with Heidelberg AppWay.”
About RetInSight
RetInSight GmbH was founded in 2020 with the ambition that longevity should not come at the expense of vision loss. We aim to enable people to maintain their full vision in order to preserve their quality of life and lifelong autonomy. We are therefore developing transformative AI-based software solutions that leverage routine optical coherence tomography (OCT) images to monitor and detect progressive retinal diseases such as GA, neovascular age-related macular degeneration (nAMD), diabetic macular edema (DME), and retinal vein occlusion (RVO) – the most important age-related diseases that lead to visual impairments in today’s world.
Our mission is to revolutionize ophthalmology through reliable, affordable, and easy-to-use AI-based medical software that enables timely diagnosis, accurate monitoring, and personalized treatment of retinal diseases for anyone, any time, and at any location by supporting networked care in ophthalmology with the help of AI technology and digital data.


